The results
The effect of resveratrol on the HeLa, U87 and HaCaT cell death ratio
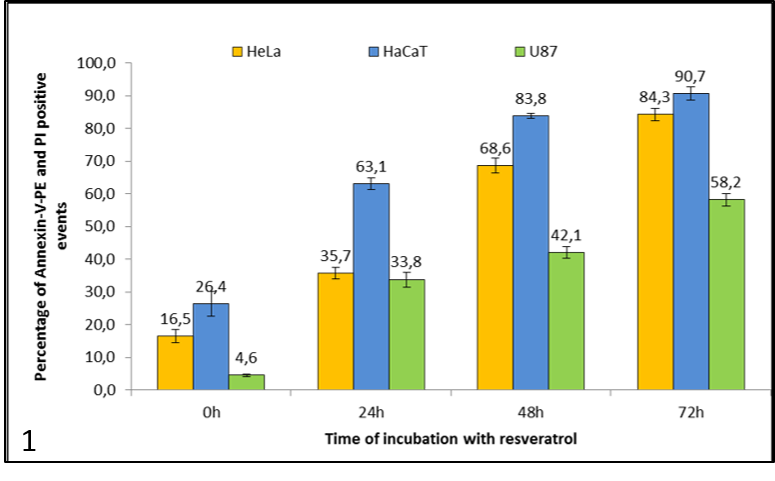
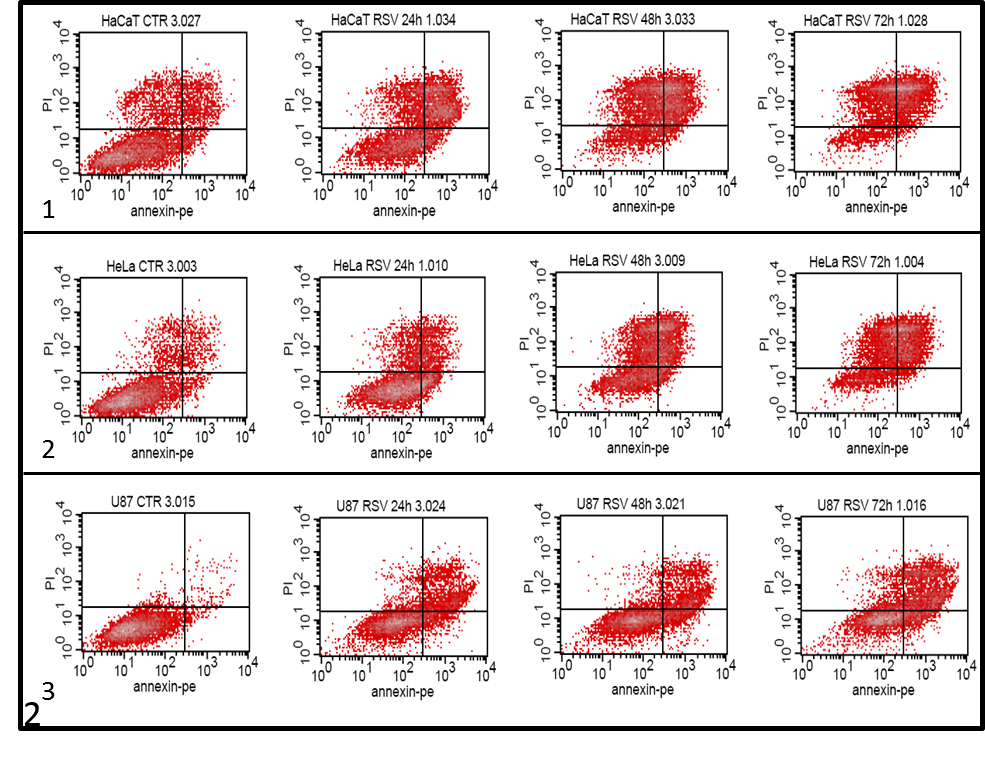
First we took a look at the amount of cell death in dependence of the time of incubation with resveratrol (graph 1 and 2 ). After treating the cells we added the colouring agents Annexin-V-PE and propidium iodide (PI) to measure the amount of dead cells with fluorescence in a flow cytometer. The dead cells are the ones in the II, III and IV quadrants in the dot plots above. An increase in the number of dead cells was noted in all treated samples, compared to the untreated cells, which rose with incubation time (graph 1). A large amount of dead cells was found in the control samples of HeLa and HaCaT cells (graph 2.1 and 2.2) in the U87 cells this percentage was almost negligible (graph 2.3). The effect of resveratrol was the fastest and largest in the HaCaT cells (graph 1 and 2.2), as over 50% of the cells were dead after 24 h. After 72 h most cells in all three cell lines were Annexin-V-PE and/or PI positive. HeLa and HaCaT cells were more sensitive to resveratrol exposure in comparison to U87 cells.
The effect of resveratrol on the morphology of HeLa, U87 and HaCaT cells
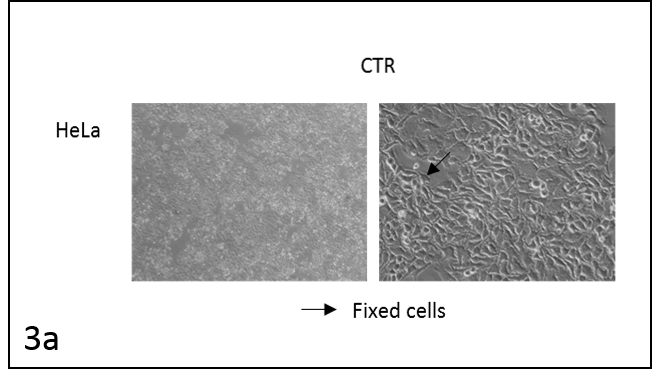
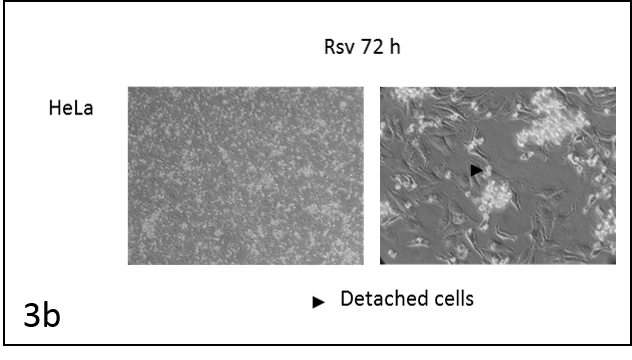
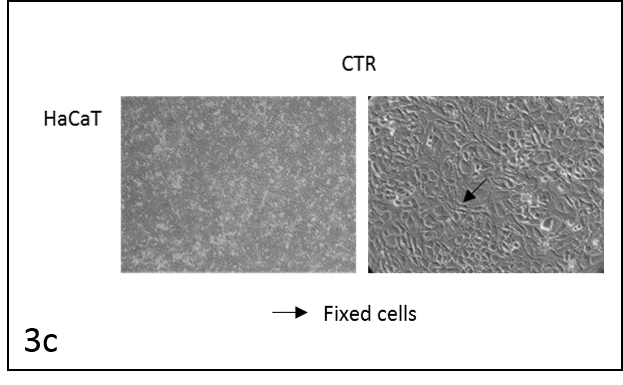
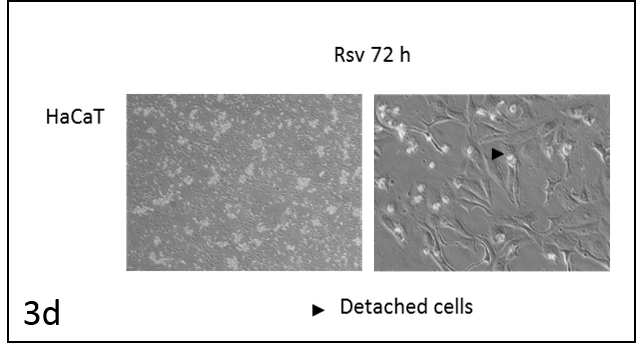

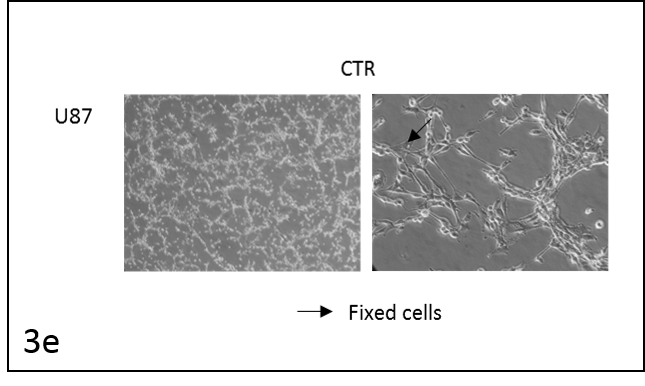
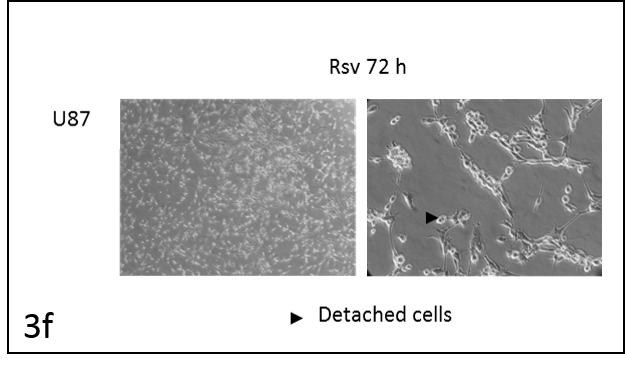
We followed the morphological changes in the cells using a microscope (pictures 3). The control cells grew normally. They were secured at the bottom of the petri dish, not many were detached from the base. The amount of detached cells after incubation with resveratrol (Rsv 72 h) compared to the control increased in all cell lines. The cells also changed shape.
Caspase activity
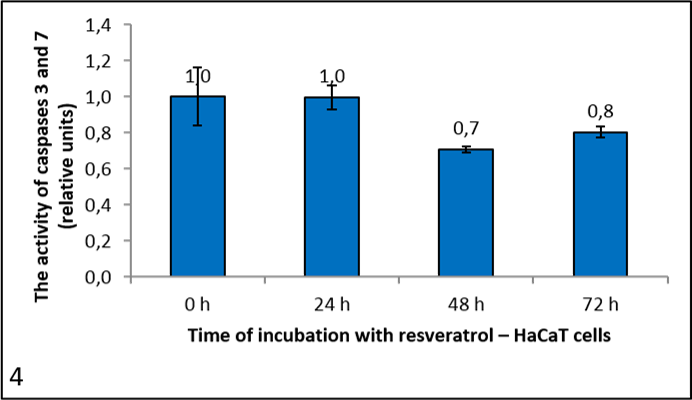
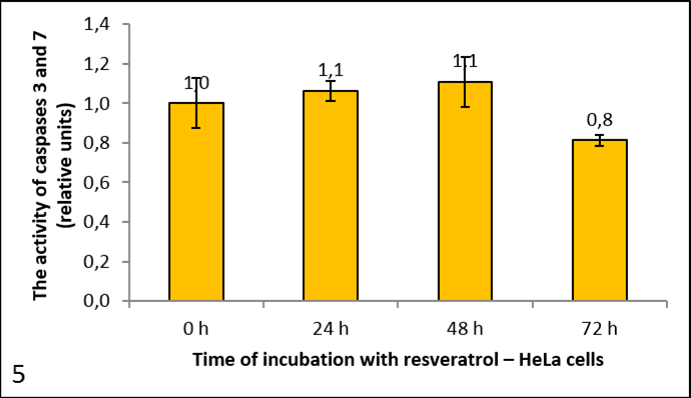
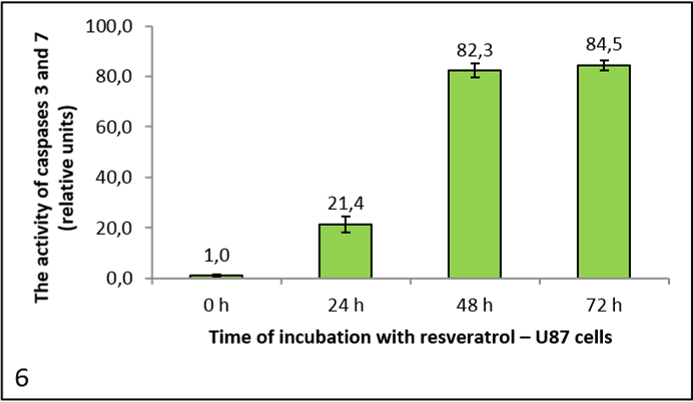
Pictures 4, 5 and 6 show the activity of caspases 3 and 7, since the substrate Ac-DEVD-AFC cleaves both caspases. It is expressed in relative units, calculated to the negative control (reference measurement). From the graph for HeLa cells (picture 5) we can see that resveratrol had no significant effect on caspase activity. It dropped after 72 h, but only negligibly. We had similar results with the HaCaT cells (picture 4). However, in the U87 cells caspase activity rose with the longer incubation times (picture 6).
Conclusion
As a result of our experiment we discovered that resveratrol effects cell death dynamic in different ways in different cell lines. The difference was a result of the different division times – HeLa cells need around 19 h for one division, HaCaT cells 24 h and U87 cells 34 h.
Through flow cytometry we proved that more cells died over time, proving our first hypothesis. For a better understanding of how the cells died we looked at morphological changes and caspase activity, since these differentiate for the two types of cell death – apoptosis and necrosis. Both results confirmed necrotic deaths in the HaCaT and HeLa cells and apoptotic death in U87 cells.
On the other hand there was no obvious difference in the effect on cancerous and non-cancerous cells, which leads us to plan new experiments in which we would test varying concentrations of resveratrol and the effect on other types of cancer. The effect on non-cancerous cells also leads to the question of how to use this substance as a target drug and limit the effect of cells surrounding tumors.
Another larger question is the difference of working simply with cells, as we do not yet know how an organism would respond to this treatment.
If this proves to be an improvement on already existing cancer treatments the distillation of resveratrol from the roots of Japanese knotweed could limit its growth and present a possible solution for this invasive plant.